Therefore the ratio between cationic and anionic radii in zinc blend is 0.39 (74pm/190 pm). The ionic radius for Zn 2+ is 74pm and for S 2-is 190pm. As we can see, in case of cesium chloride, the ionic radius of cesium is 174pm and ionic radius of chloride is 181pm which is of similar size but in sodium chloride, ionic radius of sodium ion is 102pm and ionic radius of chloride is 181pm which is not similar to each other.Ĭesium chloride is being widely used in activities such as techniques involving separation of various DNA, in analytical chemistry, medical diagnostic and treatment, etc. Since the number of atoms in a single unit cell of Zn and S is the same, it is consistent with the formula ZnS. If the size difference between the ions is large or both the ions are not similar in size then sodium chloride structure is adopted by the lattice. Structurally, the salt is a simple chloride like the more familiar sodium chloride, but because caesium is a lot closer to chlorine in size, it forms a. This can be understood as, when the size difference between the ions is small or we can say both the ions are similar in size then cesium chloride structure is adopted by the crystal. The chemical formula of cesium chloride is \ is due to size difference between the ions. On heating, crystal with cesium chloride type of crystal lattice changes to sodium chloride type of lattice.  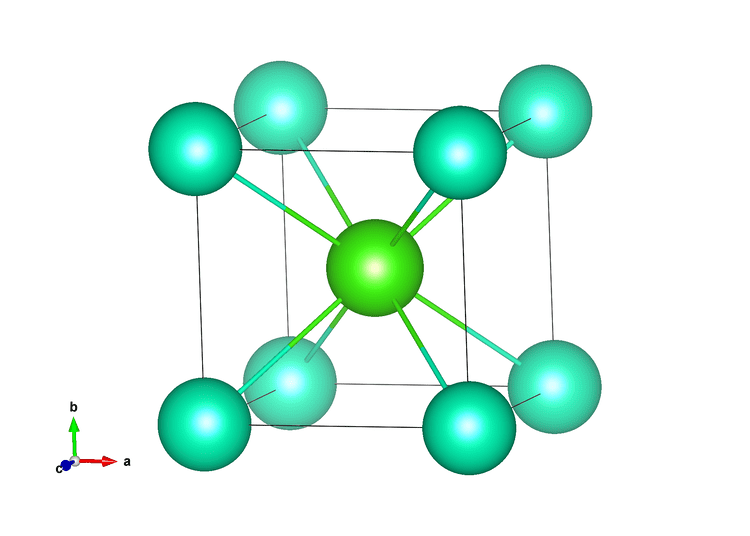
Hint:Coordination number of metal in cesium chloride is 8 and in sodium chloride type of ionic crystal, coordination number of metal is 6. Caesium chloride is widely used medicine structure in isopycnic centrifugation for separating various types of DNA.It is a reagent in analytical chemistry, where it is used to identify ions by the color and morphology of the precipitate. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
